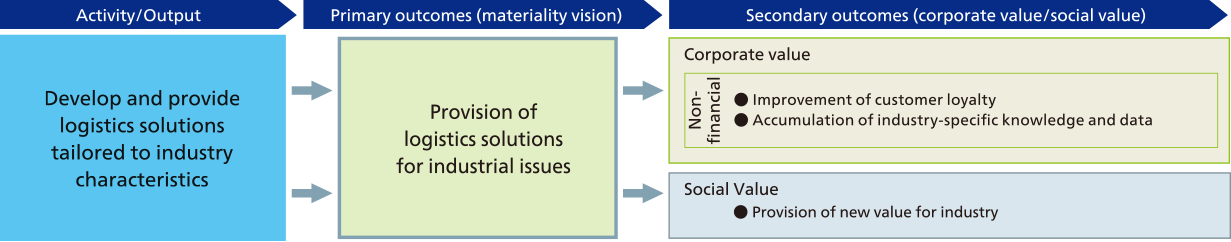
As Sustainability of the natural environment, society, and industry on which every company operates is becoming more important, it is important to provide services that not only maintain and improve the quality of services to individual customers, as we have done in the past, but also to maintain and optimize society and industry as a whole.
The NX Group aims to create new value to solve industrial issues by leveraging its strengths in advanced transportation technologies, connections with many customers in all industries and sectors, and understanding of industrial characteristics.
Pharmaceutical products require strict temperature control and security control during transportation and storage. At the same time, with the globalization of the market, counterfeiting and theft of pharmaceuticals have become major problems. Our Group aims to contribute to the health of people around the world by creating a reliable and secure global pharmaceutical logistics platform to support our customers in the global pharmaceutical industry from a logistics point of view. Therefore, we are working to obtain GDP (Good Distribution Practice for Drugs) and IATA (International Air Transport Association) CEIV Pharma certifications worldwide.
In FY2022, three bases in Japan (East Japan Pharmaceutical Center, West Japan Pharmaceutical Center, and Kyushu Pharmaceutical Center) acquired the industry’s first GDP certification for the storage and transportation of pharmaceuticals with two levels of temperature control (Room temperature: 15℃ to 25℃ , Refrigerated: 2℃ to 8℃ ). In addition, the West Japan Pharmaceutical Center confirmed compliance with Good Manufacturing Practice (GMP) standards related to pharmaceutical manufacturing.
In addition, we established the NX-Pharma Global Quality Manual in order to establish a globally standardized quality management system, and established the Pharmaceutical Logistics Global Quality Assurance Division as part of GBHQ in NIPPON EXPRESS HOLDINGS as an independent headquarters organization for quality assurance in the pharmaceutical supply network.
Acquisition of certifications for the handling of pharmaceuticals (as of March 2023)
■ Acquired GDP and CEIV Pharma certifications
at 34 offices globally located in 24 countries/regions

NX-Pharma Global Quality Manual (available in Japanese and English)
The NX Group has built a supply chain for its customers, handling everything from marine and air cargo forwarding to JIT deliveries from VMI facilities to the factory lines, from Asia to production bases around the world with a focus on Japan and Japanese automakers.
We provides VMI facility operations in various parts of the world, including the United States and China, which are the two major markets in the automobile industry, and are particularly focused on production logistics in the United States. In the United States, although the midwest has traditionally been the main region for automobile production bases, production bases are now also located in the southeast and Mexico with Japanese manufacturers already expanding their production systems. In the southeastern US and Mexico, we have newly started the operation and expansion of warehouses, and are working to expand the handling of Japanese set manufacturers and their suppliers.
In response to changes in the supply chain accompanying the shift to EVs in the industry, the NX Group is expanding its logistics network in Central and Eastern Europe, which is a production base for electric motors and lithium-ion batteries, in the European market, which is driving the shift to EVs.
Amid a drastic change in the environment, such as the outbreak of infectious diseases and CASE, automobile manufacturers have started supply chains reforms and are considering measures such as diversifying their suppliers and local production for local consumption. As a Lead Logistics Provider (LLP), in addition to providing logistics services, the Group also engages in SCM planning, design, and process management.

Please refer to the Sustainability Data Book [PDF 7,925KB] for more information on our other initiatives against this Material Issue.